
In the April issue of AQUA, the members of onBalance (Que Hales, Doug Latta and Kim Skinner) wrote a piece arguing that, for pool and spa water, a hard upper pH limit of 8.0 is overly restrictive. They claimed there should not be a wasteful mandate for closing down swimming pools when the pH is 8.0 to 8.5. We received this very well-considered response from commercial aquatics consultant Richard Young:
The April article shares some important issues about pH and cyanuric acid. The case of pH values of 8.0 having the same bacteria inactivation of water that has lower pH values has been argued for decades. There are some important points that need to be made about pH’s effect on the disinfection compound that is both wanted and needed in pool and spa water. This compound is “hypochlorous acid.”
Scientists and doctors have long recognized that the HOCl compound is the disinfecting (inactivation) agent needed to destroy viruses, bacteria and protozoan cysts (pathogens). The nature or species of HOCl is dependent upon a number of factors, as stated in the article. pH and even more CYA, as well as a slight impact by higher levels of total dissolved solids, all have an effect on what form of this chlorine compound can be found. A little basic chemistry is required here to further understand.
HOCl and CYA
HOCl is a very active compound that, by nature, is unstable. The whole idea of CYA is to “stabilize” (another name for cyanuric acid, sometimes also called conditioner) the chlorine compound, so that it is less affected by UV sunlight. It acts, if you will, like sunscreen for chlorine. The problem is that HOCl is an excellent sanitizer and oxidizer because it is unstable. When we stabilize it too much (levels of CYA above 20-30 ppm), the chlorine compound has a measurably decreased ability to do its jobs. This may be okay in backyard pools that are more “decorative ponds” but is a different story when it comes to active pools, especially public pools and most certainly spas.
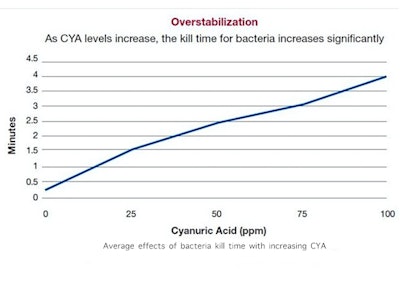 Average effects of bacteria kill time with increasing CYA (Credit: Lonza North America Water Treatment, 2011)
Average effects of bacteria kill time with increasing CYA (Credit: Lonza North America Water Treatment, 2011)
HOCl and pH
HOCl is also dramatically affected by the pH of the water. As pH decreases there are more hydrogen ions available for the OCl-(hypochlorous ions) to combine with, resulting in an increase in the formation of HOCl. As pH rises there are less hydrogen ions available and more OCl- is formed. This process is termed the “ionization of HOCI.” The definition of pH is “the inverse logarithm of the hydrogen ion content in water.”
This “ionization” concept also occurs with higher levels of TDS. The term “dissociation of hydrogen” is the term that Dr. Clifford White (often known as the “doctor of chlorination”) used. Even so, the effect of TDS on HOCl is very minor in comparison to increasing pH values.
Ionization or dissociation of hydrogen ions occurs naturally and automatically. If pH is increased, there is less HOCl available. If pH is decreased, then more HOCl forms. The graph below shows this further.
 Ionization of HOCl at various pH values (Courtesy ICC Controls, 2005)
Ionization of HOCl at various pH values (Courtesy ICC Controls, 2005)
So what does this mean for pools? At pH levels of 8.0 or even higher, disinfection still occurs to some degree. It does not take much HOCl to disinfect. That is why, years ago, chlorine residuals of 0.2 to 0.4 were acceptable within a pH range of 7.4-7.8. At chlorine levels of 1.0 to 3.0 the pH values affect the overall disinfection rate less. With all this said, it is important to discuss why the health community determined that higher levels of chlorine were necessary. Why? Oxidation.
HOCl and Oxidation
Oxidation is the “other job” many people ignore in water chemistry, but it is still very necessary. Oxidation by definition is “to remove one or more electrons from a molecule or compound resulting in a changed state.” Why is this so important? A lot more than just bacteria and viruses enter our pools and spas. Organic compounds in the form of sweat, saliva, urine and more are introduced and we need a process to destroy them. Oxidation is that process and, you guessed it, HOCl is the key.
Many have heard of “oxidation reduction potential.” A good definition is “a millivolt measurement generated by the water pulling electrons from a calibrated sensor.” To tie HOCl back into the picture, we know that it is an unstable compound. This means that it wants to take away electrons from other compounds, the result of which is that the compound breaks down or is “oxidized.” Complete oxidation reduces some compounds down to elements, such as nitrogen. ORP is used in most automatic pool chemical control systems to measure chlorine effectiveness. Not quantitative (ppm) amounts of HOCl, but qualitative, meaning that a reading of 3 ppm, free available chlorine (which measures both HOCl and OCl-) may have an ORP reading of 740 mV of ORP at 7.4 pH. Raise the pH to 8.0 and the ORP will drop from approximately 40-50 mV to 690-700 (and you can experiment this yourself if you have an ORP/pH control device). While this is still okay, if you were to add in the effects of CYA at levels above 50 ppm and or very high TDS, then trouble will be brewing (pathogens too!).
The World Health Organization recognizes that 650 mV of ORP is the lower limit of effective pathogen inactivation. As pH increases, studies have clearly shown that ORP drops. As pH decreases ORP rises, and ORP is then directly tied into the rate of pathogen destruction.
The higher the concentration of HOCl in the water, the higher level of oxidation of organic compounds occurs. Failure to destroy those compounds results in HOCl compounds combining with organic compounds to form combined chlorine, otherwise known as “chloramines,” of which there are several species. These compounds result in odor and eye burn.
So what does all this mean? Higher pH levels result in ionization of HOCl and the concentration of OCl- increases. OCl has significantly lowered ORP value and, as a result, the inactivation of pathogens is slower, but I might add, in most cases, still occurs. In addition, scientists have completed studies of bacteria inactivation at various ORP values. The higher the ORP values, the more quickly pathogens are inactivated. The World Health Association recognized 650 mV of ORP is the lower limit to achieve adequate pathogen inactivation. That would be around 1 ppm free chlorine in 8.8 pH water.
We will not even begin to discuss the water balance issues that can arise with increasing pH, but of course, there are challenges there, too.
To wrap things up, ORP is the best indicator of pathogen inactivation (there are several other studies that could be referenced). Oxidation is the best way to destroy organic compounds. ORP is the measurement of oxidation potential. Impacting the ORP downward decreases the speed in which pathogens are destroyed in addition to decreasing the water’s ability to destroy organic compounds. If you operate a busy pool and want to keep out of trouble, keep your pH in the mid 7s, your CYA at zero to a maximum of 20 ppm and maintain a free chlorine residual of 2-3 ppm. But if you have a decorative pond, you have a lot more room for variations.
Rich Young has over 35 years of municipal, public and commercial swimming pool and water park industry experience. He founded Aquatic Commercial Consulting in 1992 and is presently the principal of this independent aquatic facility consulting firm located in Saratoga, Calif.











































