
In the swimming pool industry, one of the most popularly chosen forms for sanitizing and superchlorinating water is sodium hypochlorite. Commonly known as "liquid chlorine" or bleach, sodium hypochlorite is widely used in both commercial and residential swimming pools. Sodium hypochlorite effectively destroys bacteria and prevents algae in swimming pools.
This edition of Tech Notes provides information on the characteristics, effects and proper application of sodium hypochlorite.
SODIUM HYPOCHLORITE: THE BASIC FACTS
- Clear yellow liquid with a chlorine odor
- A solution containing water, hypochlorite, sodium hydroxide and a trace amount of sodium chloride
- Strong bleaching agent
- Effective sanitizer for most pathogens
- Prevents and kills algae
- Oxidizes ammonia and swimmers' waste
- Provides a measurable chlorine residual in the water
WHAT IT IS
Sodium hypochlorite (NaOCl), commonly referred to as "liquid chlorine" or liquid bleach, is an aqueous solution created by mixing chlorine gas in water with concentrations of sodium hydroxide. This liquid is manufactured at differing trade percentages.
Household bleach for general cleaning purposes and laundry is available in concentrations from 1.5% to 6% sodium hypochlorite, by weight. Sodium hypochlorite for swimming pools is usually manufactured at concentrations of 10% or 12.5%. One gallon of 12.5% sodium hypochlorite provides approximately 12.5 ppm of free chlorine per 10,000 gallons of pool water. It takes 10.6 fl. oz of 12.5% sodium hypochlorite to get roughly 1 ppm of free chlorine in 10,000 gallons of pool water. The pH of pool grade sodium hypochlorite is 13.
RELATED: Pool Sanitizing: A Return to the Simple
Sodium hypochlorite is classified as an inorganic sanitizer; it does not contain cyanuric acid. Sodium hypochlorite is a primary sanitizer because of its ability to kill microorganisms, oxidize nonliving contaminants like ammonia and swimmers' waste and provide a protective residual in the water. Sodium hypochlorite is non-flammable, non-combustible and non-explosive, and containers under 1.3 gallons are transported as "Limited Quantities" with no transport hazard classification.
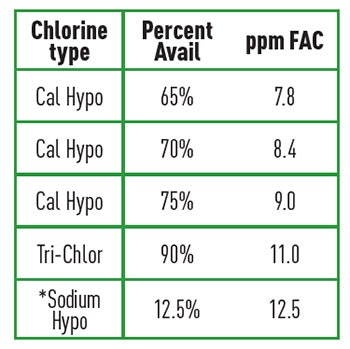
Containers over 1.3 gallons should be labeled for transportation as UN1791, hazard class 8. Table 1 (on page 79) shows the ppm of free available chlorine in 10,000 gallons from 1 gallon (10.1 lbs.) of liquid* compared to 1 lbs. per 10,000 gallons of solid chlorinating compounds. Sodium hypochlorite is classified by the Uniform Fire Code as a corrosive. As such, a maximum of 500 gallons can be stored in a non-fire, sprinkler-protected room and 1,000 gallons in a fire, sprinkler-protected room as maximum exempt quantities. Quantities beyond this create an "H" Hazardous Occupancy and require special fire protection.
Sodium hypochlorite reacts in water to create hypochlorous acid (HOCl). HOCl is the most effective sanitizing chlorine species available. HOCl disassociates to OCl- (hypochlorite anion), and the amount of disassociation is based on pool water pH. At a target pH of 7.5, 50% of the chlorine exists as HOCl and the other 50% as OCl-. Since HOCl is the killing agent form of chlorine, it is important that pool water pH be balanced to the most ideal target levels. Because of its aqueous solution, sodium hypochlorite is immediately available to provide HOCl. It is also an oxidizer that removes non-living contamination and is one of the preferred shocking methods, since high doses will not contribute to the increase of calcium hardness or cyanuric acid levels. Sodium hypochlorite is safe for use on all types of pools including plaster, colored plaster, aggregates, vinyl liners and fiberglass.
WHAT IT DOES
Sodium hypochlorite loses strength over time as it is converted to sodium chloride and water. Large stock quantities should be discouraged. It is better to purchase and store sodium hypochlorite in smaller quantities that encourage inventory turnover. The rate of decline of sodium hypochlorite's strength is influenced by physical and chemical conditions such as high temperature, exposure to UV light, initial concentration and purity of the product. Sunlight and high temperature can speed up the process. Sodium hypochlorite (12.5%) will reach its halflife in approximately 180 days when stored at a temperature of 77 °F/25 °C. At 95 °F/35 °C the half-life is 48 days. The presence of metal contaminants or impurities in the manufactured product or storage tank will increase the degradation rate. It is important to keep sodium hypochlorite stored in cool, dark and well-ventilated storage areas when possible. Filtered sodium hypochlorite has a longer shelf life.
Pool grade U.S. Environmental Protection Agency (EPA) registered sodium hypochlorite has a pH of approximately 13. With its use, the degree to which it increases total dissolved solids (TDS) can vary.
HOW TO USE IT
Sodium hypochlorite can be applied manually or with use of a positive displacement feeder such as a diaphragm, peristaltic pump or a venturi feeder system. Manual addition should be done in the deep end of the pool away from any metal parts such as rails, ladders or light rings. Pour the liquid very slowly while holding the container as close to the water surface as possible. Pouring from a high position will lead to undiluted splash back of product on decks and clothing, which will cause bleaching. It is recommended to wear protective gloves and goggles when applying. Sodium hypochlorite feeder pumps should be set up with the injection point after all equipment to prevent undiluted chemical contact with pumps, filters and heaters. Feeder pumps should be calibrated in accordance with the manufacturer's specifi cations, and tubing should be checked regularly for brittleness and leakage. Feeders should be installed to avoid siphoning.
PRECAUTIONS
- Sodium hypochlorite is corrosive and should be stored in a cool, well-ventilated storage area away from metals and other chemicals.
- Care should be taken when applying to prevent contact with eyes and skin. Use gloves and eye protection such as goggles or a face shield.
- Only U.S. EPA registered sodium hypochlorite products should be used.
- Sodium hypochlorite products should be used according to the manufacturer's label instructions.
- The filtration/circulation system should be running when adding products.
- Bathers should not enter the pool until the tested chlorine levels are at 4 ppm or less.








































